
Teal Health Teams Up With Thatch to Expand Access to Cervical Cancer Screening
Through a new partnership, Thatch’s employer customers can now offer Teal Health’s at-home cervical cancer screening as an employee benefit through an ICHRA.

Through a new partnership, Thatch’s employer customers can now offer Teal Health’s at-home cervical cancer screening as an employee benefit through an ICHRA.

HPV self-collection testing empowers more people to participate in their own care, fostering autonomy and helping to bridge long-standing gaps in access to screenings for all populations.

Teal Health received FDA approval for the first-ever at-home cervical cancer test, which seeks to give patients an alternative to the in-office pap smear. The test is already covered by four major payers in California.
Teal Health raised $10 million in seed funding, taking its total funding to $23 million. The company has submitted an application to the FDA for its at-home cervical cancer screening kit, which seeks to give patients an alternative to the in-office pap smear.
The Seagen and Genmab antibody drug conjugate (ADC), named Tivdak, is approved to treat cases of cervical cancer that have returned or spread following treatment with chemotherapy. Tivdak is Seagen’s third ADC; for Genmab, the drug will become its first commercialized product.

It is unclear why the American Cancer Society would potentially impact the lives of countless women with recommendations that contradict decades of real progress.

Enterprise EHR boosts scalability, interoperability, and governance for large healthcare systems.

The company said the agency told it that it may consider data from an amended version of the trial of LN-145 for approval. An analyst wrote that approval could happen in the first half of 2021, based on Iovance's guidance for regulatory submission.
The Series A financing round of $2.25 million will be used to move forward with the development of its point-of-care diagnostic tools for the accurate detection of epithelium cancers, with a primary focus on cervical cancer.

Genentech, whose drug Avastin is approved to treat advanced cervical cancer, has launched a contest offering $50K to startups with innovative approaches to address the disease.

San Diego startup Hera Therapeutics is developing a topical antiviral drug that attacks human papillomavirus – and early studies are showing it can successfully combat three strains, including two that cause 70 percent of all cervical cancer. The drug candidate, HTI-1968, blocked the replication of HPV-16, HPV-18 and HPV-11 cells, according to the NIAID-sponsored study. The work […]

Small practices play a critical role in healthcare delivery, but they cannot continue to absorb ever-increasing administrative demands without consequences.

You never want to be the last speaker at the end of a long conference day. Many people in the 4:30 slot even mention this strategic disadvantage, “I know I am standing between you and happy hour, so I’ll make it quick.” The best end-of-the-day speaker I have ever heard was Dr. Victor Montori at […]

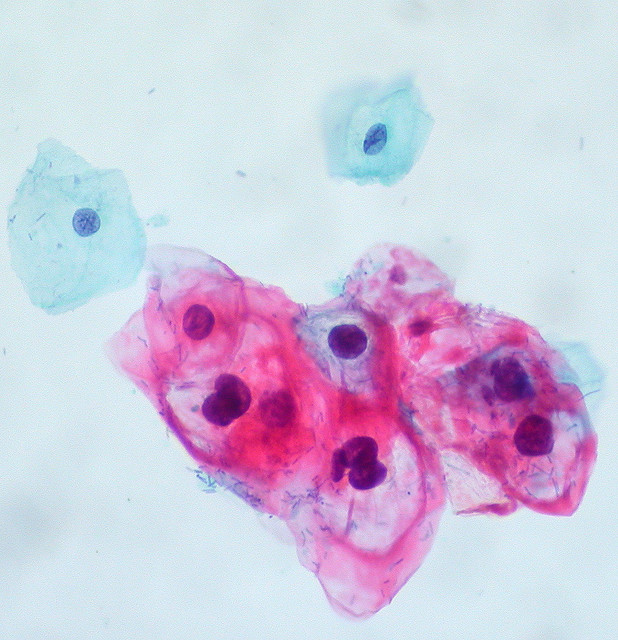
Cervical cancer is the easiest female cancer to prevent, because there are highly effective vaccines available, and precancerous cells can be detected by Pap tests and treated before they turn into cancer. Yet, in some areas of the world with lower incomes, cervical cancer incidence rates are still high, partially because of limited access to […]